Phase 1
The tracer and process map (Fig. 1) identified key steps and decision points in the old ICBP process. The process started with the initial decision to scope made by the GI team which were comprised of fellows and attendings and were the same mix of experience levels throughout our entire study. Next, the primary team (residents, attendings, and advanced practitioners)placed orders, not consultants. Several steps performed by the patient, physicians, nurses, and pharmacists are performed on the day prior to the procedure and the morning of the procedure.
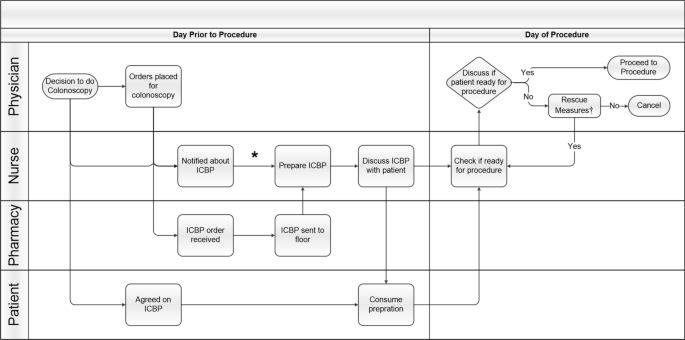
Inpatient Colonoscopy Bowel Preparation Process Map. Steps are shown at each level of care between physician, nurse, pharmacy, and patient from decision to do colonoscopy to the procedure. Abbreviations: Inpatient Colonoscopy Bowel Preparation (ICBP). *Units that perform ICBP frequently have ICBP stored in supply room on their unit. †Rescue measures: suppositories, enemas, additional bowel preparation
In the cause-effect diagram (Additional file 1: Fig. 1), causes for a poor ICBP based on our old process were placed into 6 categories: patient, supply, order, timing, provider, and communication. For example, we determined that orders for procedures were being placed late in the day due to late communication from GI to primary team of the procedure. Therefore, the patient was drinking the bowel preparation late into the evening. Another problem identified was patient education about the ICBP process was based solely on verbal communication as opposed to having supplemental visual aids. We found high variability in bowel preparation recommendations with the predominant approach being a single dose of 4 L the night before procedure. We determined that palatability was a problem because juices and powders used to improve the flavor of the solution were used infrequently because they were difficult to obtain by nurses. Lastly, we uncovered there was no standardization in the use of nasogastric tubes and in the time of day inaptient colonoscopies were scheduled.
Phase 2
During the study phase of each PDSA cycle, we performed additional tracers and iterative discussions with patients and front-line providers at various points. For example, we engaged with the front line to discuss when an ICBP was performed on a floor where ICBPs are infrequently performed. These PDSA cycles allowed us to address key aspects of ICBP that were previously unspecified, such as nursing and patient concerns, tolerance of nasogastric tubes, and anti-emetic use. We developed several solutions described in detail below that contributed to our final protocol.
Early verbal communication by 4 p.m. of decision to scope to the primary team allowed time for pharmacy to deliver preparation medications and for nursing to prepare the medication. Since prior studies have demonstrated inpatients have a higher incidence of poor ICBP [2, 5, 6, 10, 12, 14] and guidelines recommend 6L bowel preparation for acute lower GI bleeding [32], our protocol included a 6L split-dose regimen with polyethylene glycol-electrolyte solution (PEG-ELS) with dose adjustment as needed. Based on recent publications, the use of polyethylene glycol (PEG) 3350 was phased out of practice at our institution and a sulfur-free version was added to formulary [18, 32, 33]. The ICBP start time for the first dose of 4L was between 4 p.m. and 6 p.m. the night prior to procedure at a rate of 1L per hour which allowed the patient to sleep. The second dose of 2L started at 5 a.m. and completed by 7 a.m.. Nurses used toilet specimen collectors to accurately assess stool for adequacy of ICBP. Enemas and suppositories were previously used as “rescue” in the morning were phased out of practice as they did not reach the right colon. Following evidence-based anesthesia recommendations [1, 34, 35], the inpatient colonoscopies were scheduled for a start time at 10 a.m. which allowed 2–3 h after completion of the morning dose.
We addressed communication between GI and primary team using a simple IT solution. Electronic Health Record (EHR) note templates were placed prominently in the recommendations section of the consultation note to streamline clear communication to the primary team and nursing. The templates included primary team and nursing instructions, recommendations for anti-emetics, steps to improve palatability, and the use of nasogastric tubes, where appropriate. Since the use of nasogastric tubes is recommended per guidelines for high volume bowel preparation [32], their use at the discretion of the GI and primary team was determined a priori.
ICBP education was provided to patients, nurses, and physicians in the form of information sessions, point-of-care training, and handouts. Using the Pareto Principle, an engineering concept that a small percentage of participants constitute the majority of the action of interest, we identified the few nursing units and primary teams performing the most ICBP. We initially targeted these units and teams with education sessions, and these sessions were later expanded to include most other residents and nursing units in the hospital. Our patient education handout was described the importance a good ICBP, pictures, and frequently asked questions (Additional file 2: Fig. 2) [36]. Figure 2 shows the final protocol which includes an example of templated text in the electronic health record.
Gastroenterology Inpatient Bowel Preparation Protocol. GI: gastroenterology; BBPS: Boston Bowel Preparation Scale, PEG-ELS: polyethylene glycol-electrolyte solution
There were 120 and 129 bowel preparation episodes in 119 and 128 patients in the pre- and post-intervention groups, respectively. Table 1 shows the patient characteristics in the pre- and post-intervention periods. The individual factors of age, sex, race, ethnicity, primary team of internal medicine, and indications for procedure were similar between groups. The most common indication was bleeding. The system factor of hourly bed occupancy was a measure of how busy the hospital was in the week of ICBP and was statistically significantly higher in the post-intervention group. Therefore, we adjusted for this in the regression model.
We calculated the percentage of planned colonoscopies per week that resulted in a good ICBP for the pre-intervention period. The median percentage of good ICBP for the pre-intervention period was identified as our baseline which was 66.7%. We used this baseline to compare our post-intervention period. Each week, we evaluated our intervention through PDSA cycles using the new data as a guide. Figures 3, 4 shows a run chart of the percentage of good ICBP by week for the pre- and post-intervention periods. A “shift”, 6 or more consecutive points above the baseline [29], is seen from week 18 to week 25. One-point deviations, such as the one seen in week 26, are considered noise in the process due to the stochastic nature hospital processes, so the focus is on trends. Looking overall at this run chart, one can appreciate the improvement in good ICBP and increased consistency in the post-intervention period.
Run chart of percent good inpatient colonoscopy bowel preparations with pre-Intervention baseline to compare with post-intervention. ICBP: Inpatient Colonoscopy Bowel Preparation. *Represents time period for development and deployment of intervention between pre and post-intervention periods
Inpatient Bowel Preparation Quality Improvement Methodology. Outline of our methods using Health Systems Engineering principles and the Model for Improvement to improve inpatient bowel preparations. GI: gastroenterology
Prior to intervention there was no statistically significant time trend (the percentage of adequate ICBP was not increasing or decreasing) which demonstrated the intervention was not following an already upward trajectory of improving ICBP. Similarly, we did not observe statistically significant time trend in the post-intervention period which demonstrates stability of the impact of the intervention over time. For example, if we had found a downward trend in ICBP in the post-intervention period, we would be concerned the intervention effect was not sustained. Thus, below we report the results of the models with intervention as the primary predictor to evaluate the impact of the intervention. In addition, since the pre and post periods had different occupancy levels, this variable was added to the model for adjustment.
Our regression model showed the pre-intervention percent of good ICBP was 61% compared to 74% after the intervention with estimated odds ratio of a good ICBP comparing post- to pre-intervention of 1.87 (95% CI 1.09–3.21, p value = 0.02). The estimated odds ratio did not substantially change after adjustment for average weekly occupancy (Table 2).
The regression analysis showed that before the intervention the percent of ideal ICBPs was 53% compared to 69% after the intervention. The estimated odds ratio of ideal ICBP comparing post- to pre-intervention was 1.95 (95% CI 1.16–3.27, p value = 0.01). The estimated odds ratio of ideal ICBP did not substantially change after adjustment for average weekly occupancy. There was estimated 25% decrease in LOS in the post intervention compared to pre-intervention period, from mean of 8 days in the pre-intervention period to mean of 6 days in the post-intervention period. This decline, however, did not reach statistical significance (95% CI 0.54–1.07, p value = 0.1).
link

