Voyxact gets accelerated approval, offering investors a new entrant in the high-value kidney disease market. The Food and Drug Administration...
FDA
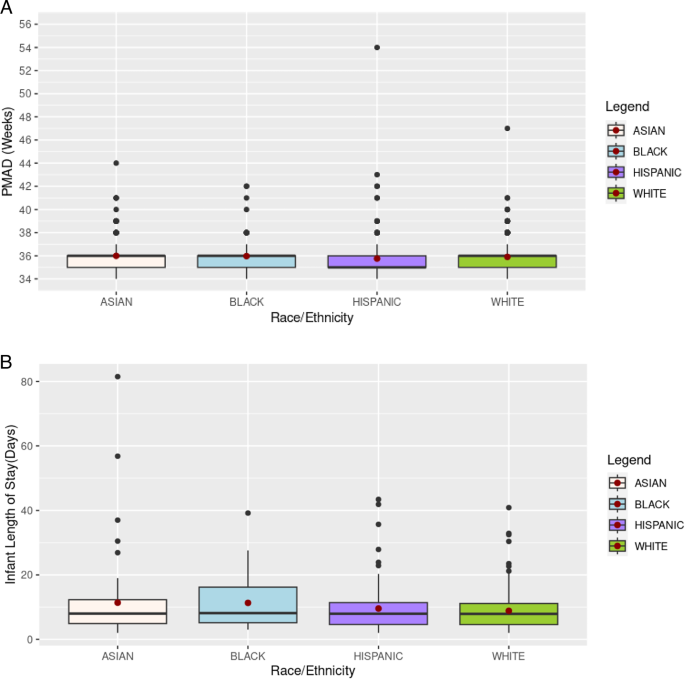
The US FDA is warning consumers, caregivers, and healthcare providers of risks related to using unauthorized infant monitors for measuring...
A Travere Therapeutics drug for a rare disease that can progress to kidney failure has converted its status to full...